THE EXCEPTIONAL DIFFERENCE
Although Cure Medical intermittent catheters function like others, they offer unique features which translate into benefits that our customers value:

Cure Medical manufactures intermittent catheters in a variety of styles, sizes, and coating options.
NOT MADE WITH DEHP/DINP, BPA, OR NATURAL RUBBER LATEX
 Cure Medical products are not made with DEHP/DINP, a known carcinogen, not made with BPA, and not made with Natural Rubber Latex, a common allergen.
Cure Medical products are not made with DEHP/DINP, a known carcinogen, not made with BPA, and not made with Natural Rubber Latex, a common allergen.
A study conducted at Craig Center in Denver found that people with SCI are 16 to 28 times more likely to get bladder cancer. To finally have a product on the market that is not made with cancer-causing plasticizer or natural rubber latex is a huge advancement. Learn more about the concerns with DEHP here.
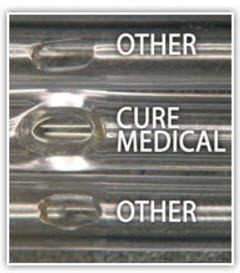
High magnification Scanning Electron Microscope (SEM) imaging of an eyelet on two national brand catheters and a Cure Medical catheter reveals differences in quality that can be seen and felt.
ALWAYS SMOOTH, FIRE-POLISHED EYELETS
Unlike many PVC catheters that are cold-punched in the manufacturing process, all Cure Medical catheters have smooth, fire-polished eyelets for increased comfort and a more positive catheterization experience.
POPULAR STYLES AND SIZES
Cure Medical offers a variety of styles and sizes to help meet the needs of any catheter user. See our ready-to-use, pocket-sized, pediatric, and closed systems for example. View all catheters here.
GREAT VALUE
Although Cure Medical products are of exceptional quality, they are still ‘reimbursement friendly’.
